Protons neutrons and electrons are the three main subatomic particles found in an atom. The atomic number of an atom is given by the number of protons present in the atom.
What Are The Subatomic Particles In An Atom Including The Charge Location And Mass Of Each And Their Relationship Within The Atom Socratic
The other elementary particles are mediators of the fundamental forces.
Which subatomic particles are charged. The number of protons in an atom is equal to the number of electrons in it. Subatomic particles include electrons which are the negatively charged almost massless particles which nevertheless account for most of the size of the atom. There are many different types but three of the most common are protons neutrons and electrons.
Subatomic particles are particles that are smaller than the atom. Every atom has a specific set of identical protons and identical neutrons. 6 1 0 1 9 Coulomb Electrons are extremely small and have a mass of about 11837 the mass of a proton or neutron.
By breaking them down even more subatomic particles are often made up of elementary particles. These mediator particles enable the matter particles to interact with each other. Protons have a positive charge.
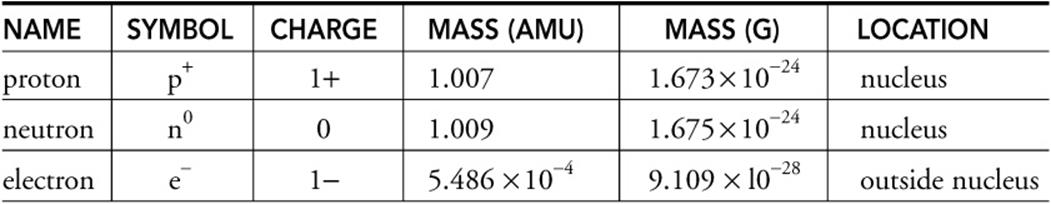
All the positive charge of an atom is contained in the nucleus and originates from the protons. Atoms are neutral and uncharged even though they contain charged subatomic particles. The mass of the neutron nearly equals the mass of a proton.
The proton was discovered by Ernest Rutherford in the year 1919. Protons can be produced via the removal of an electron from a hydrogen atom. Electrons are negatively charged and are the heaviest subatomic particle.
Protons are positively charged and the lightest subatomic particle. Relative Charges of the Proton Electron Neutron Protons neutrons and electrons are the subatomic particles that make up atoms. The atom is the smallest particle of matter that cannot be divided by chemical means but atoms are made up of smaller particles called subatomic particles.
All observable subatomic particles have their electric charge an integer multiple of the elementary charge. Protons lie in the nucleus of the atom. Baryons and mesons are combinations of quarks and are considered subatomic particles.
The third subatomic particle electrons are the negatively charged particles with charge of - 1. The three main subatomic particles of an atom are protons neutrons and electrons. Protons neutrons and electrons are the three main subatomic particles found in an atom.
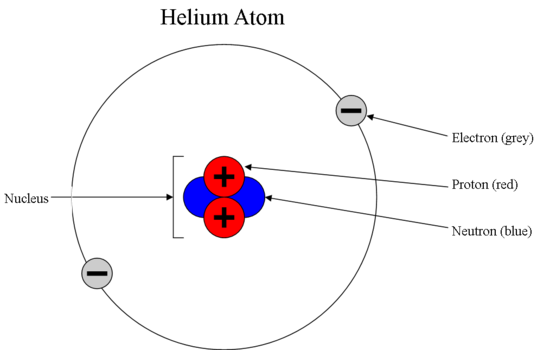
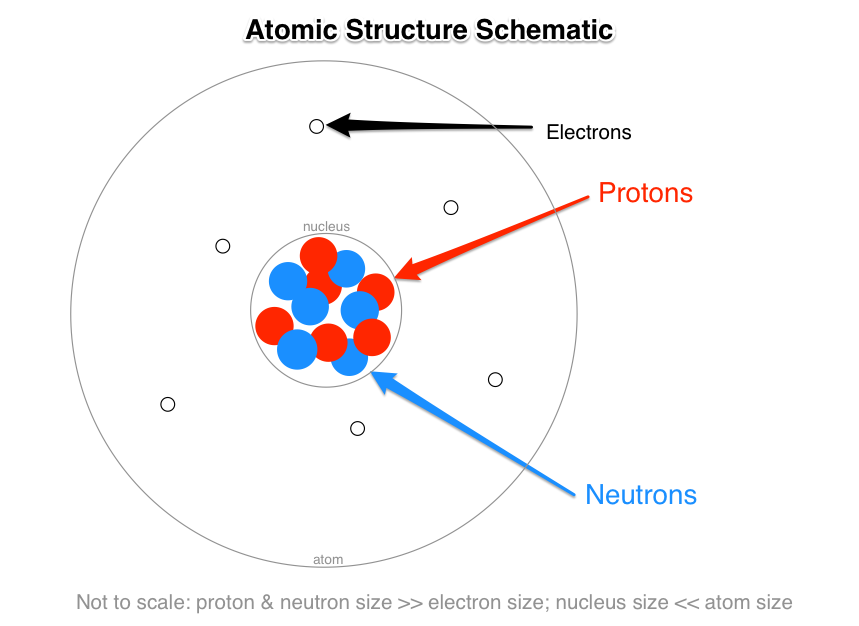
The subatomic particles of protons and neutrons are found in the nucleus of an atom. The Bohr model shows the three basic subatomic particles in a simple manner. Most of an atoms mass is in the nucleus a small dense area at the center of every atom composed of nucleons.
And the answer is Yes. An easy way to remember this is to remember that both p roton and p ositive start with the letter P. It includes the heavier building blocks of the small yet dense nucleus of the atom the positively charged protons and the electrically neutral neutrons.
Neutrons have no electrical charge. An atom is made up of about 200 different types of particles such as proton neutron electron muon positron tau and blah blah blah. The Standard Models quarks have non-integer electric charges namely multiple of 1 3 e but quarks and other combinations with non-integer electric charge cannot be isolated due to color confinement.
Mats Persson Getty Images The atom is the smallest particle of matter than cannot be divided using a chemical means but atoms consist of smaller pieces called subatomic particles. Protons are positively charged subatomic particles. Protons are particles with a positive charge while neutrons have no charge.
Subatomic particles include electrons the negatively charged almost massless particles that nevertheless account for most of the size of the atom and they include the heavier building blocks of the small but very dense nucleus of the atom the positively charged protons and the electrically neutral neutrons. Neutrons have no charge and are the lightest subatomic particle. Protons have a positive charge.
Subatomic particles are particles that are smaller than an atom. The mass of an atom is concentrated in the nucleus as the nucleus contains the heaviest subatomic particles the neutrons and protons The mass of the electron is negligible The nucleus is also positively charged due to the protons. It is a positively charged subatomic particle with a mass of 16726 10-27 kg which is 1836 times that of an electron.
The discovery of protons is credited to Ernest Rutherford. The most famous baryons are protons and neutrons. Each of the subatomic particles has a different mass and a different electric charge which is summarised in the following table.
These are called subatomic particles. Electrons which have a negative charge are particles that can found orbiting outside the nucleus of an atom. Nucleons include protons and neutrons.
The three main subatomic particles of an atom are protons neutrons and electrons. Electrons have negative - charge.

2 1a Overview Of Atomic Structure Biology Libretexts
Learn Mass And Charge Of Subatomic Particles In 2 Minutes

Subatomic Particles Definition Discovery And Key Features
What Are The Names Charges And Locations Of The Three Types Of Subatomic Particles That Make Up An Atom Socratic
